Author: Jean-Michel Arnal, Simon Franz
Date of first publication: 16.02.2018

Spontaneous breathing trials (SBTs) are among the most commonly used techniques to facilitate weaning from mechanical ventilation. Conducting the initial SBT with pressure augmentation was more likely to be successful, produced a higher rate of extubation success and was associated with a trend towards lower ICU mortality when compared with the T-piece SBT (
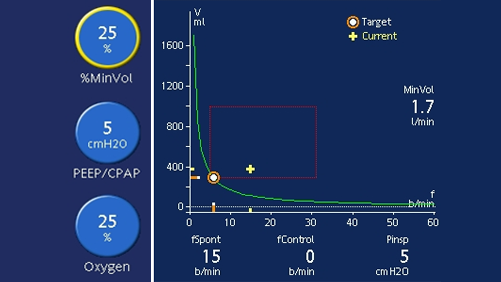
If your patient is on ASV, you can perform a pressure-support SBT as follows. Firstly, decrease PEEP to 5 cmH2O and lower the target minute volume (%MinVol) setting to reduce the pressure support as needed. If the pressure support on the current %MinVol setting is higher than 15 cmH2O, the %MinVol setting can be lowered first to 70%, and then to 25% to gradually reduce the level of pressure support down to 5-8 cmH2O for the SBT.

The patient should be monitored both clinically and using the Vent Status panel. Clinical monitoring includes neurological status, effort to breathe, heart rate, and blood pressure.
Respiratory monitoring includes SpO2, PetCO2, respiratory rate, and tidal volume. Patient exhaustion may lead to an increase in the respiratory rate with a small tidal volume, and cause an increase in pressure support according to the ASV algorithm.
If the patient remains stable for 30 minutes, you can consider extubation (
Relevant devices: all