Author: Aude Garnero, Intensivist, Hôpital Sainte Musse, Toulon, France
Date of first publication: 04.05.2021
High flow therapy (HFT) has been used widely during the COVID-19 pandemic. Published clinical experience is based mainly on retrospective studies and describes the main settings and the failure rate.

In these studies, the flow setting at the start of HFT was between 30 L/min and 60 L/min (1-9). Oxygen was between 60% and 100% at initiation (
The temperature was set to between 31°C and 37 °C (
Flow is usually set to cover the patient’s minute volume ventilation, and is then adjusted according to the patient’s tolerance. Oxygen is adjusted dynamically based on SpO2 values and blood gas analysis. The temperature is adjusted according to the patient’s comfort, but the higher the temperature, the higher the humidity provided.
1. When to start HFT?
If a COVID-19 patient remains hypoxemic despite conventional oxygen therapy, recommendations suggest using HFT over conventional oxygen therapy (
2. Initial settings:
3. Adjustments:

4. How to wean from HFT?
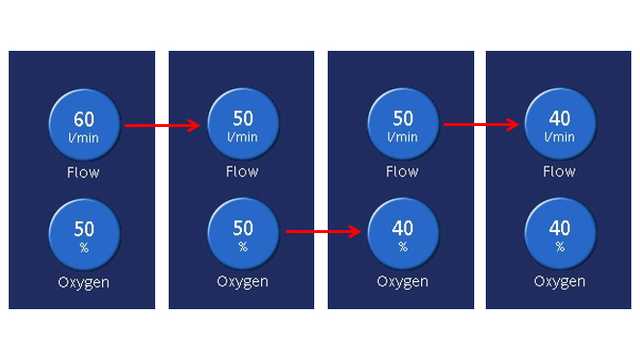
When Oxygen is higher than 50%, HFT can be combined with prone positioning or CPAP.
Full citations below: (

Our e-book on high flow nasal cannula therapy gives you an overview of the working principles and clinical benefits, as well as practical information about choosing the right interface, adjusting the settings, and monitoring your patients.