Author: Jean-Michel Arnal, Senior Intensivist, Hopital Sainte Musse, Toulon, France
Date of first publication: 19.10.2018
Last change: 09.09.2020
FormattingA recent physiological study demonstrated that esophageal pressure estimates the pleural pressure at mid-thorax at all levels of PEEP. Therefore, an absolute measurement of esophageal pressure is useful for setting PEEP and monitoring transpulmonary pressure.

So how do we measure esophageal pressure correctly?
The esophageal balloon has to be positioned and inflated correctly, and placement then verified.
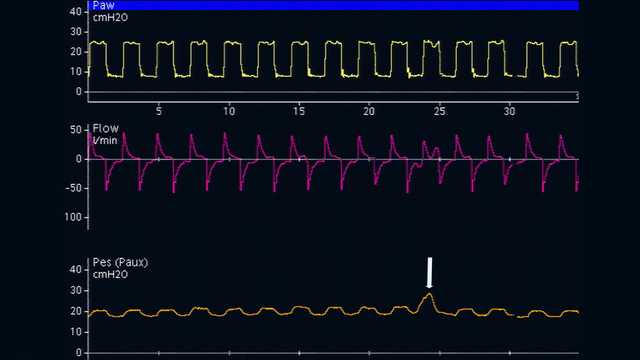
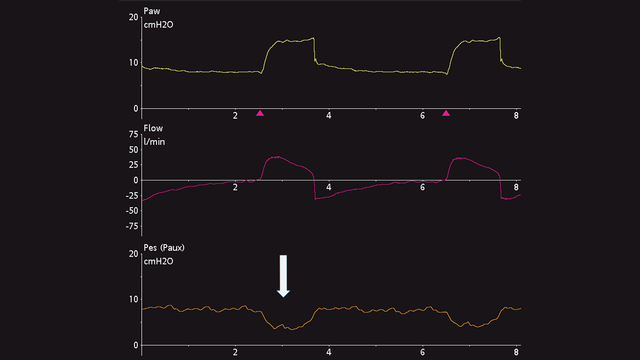
The optimal position for the esophageal balloon is the lower third of the esophagus, at a distance of 35–45 cm from the nostrils. With a patient in semi-recumbent position, the empty balloon is first inserted into the stomach, which is situated around 50–60 cm from the nostrils. The balloon is inflated to a standard volume (1 ml for a Cooper Surgical catheter and 4 ml for a Nutrivent catheter). Gastric pressure displays a positive deflection during inspiration in both passive and spontaneously breathing patients. The gastric position is ascertained by gently applying manual epigastric compression, which shows an immediate increase in gastric pressure (see Figure 1).

The esophageal catheter is then gently withdrawn while the balloon is still inflated, in order to position the balloon in the lower third of the esophagus. During the change from gastric (see Figure 2) to esophageal (see Figure 3) pressure, the baseline of the pressure waveform changes and cardiac oscillations appear.
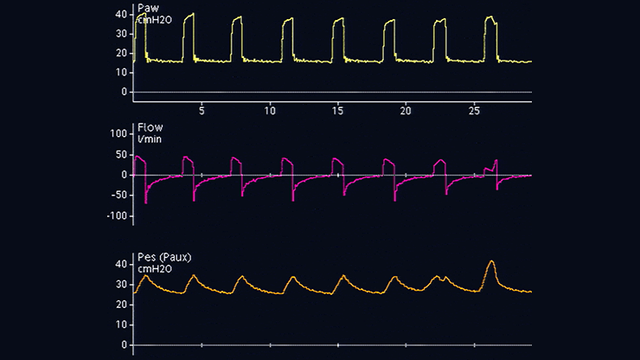
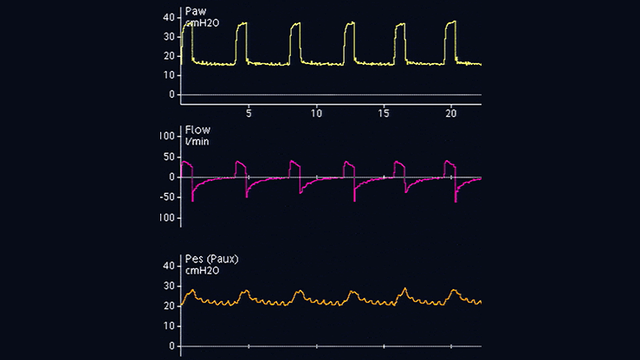
Esophageal pressure deflections are positive during inspiration in passive patients (see Figure 4), but negative in spontaneously breathing patients (see Figure 5). If cardiac oscillations distort the esophageal pressure signal, you can withdraw the catheter by another 2–5 cm.
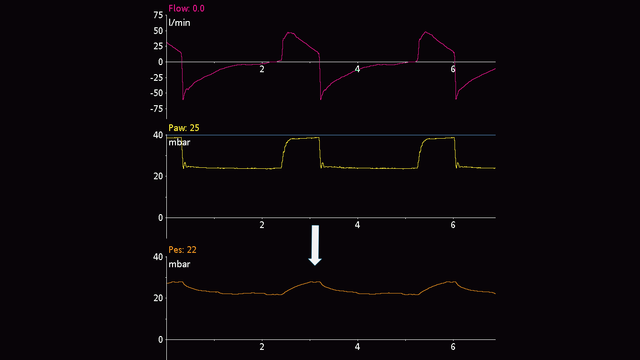
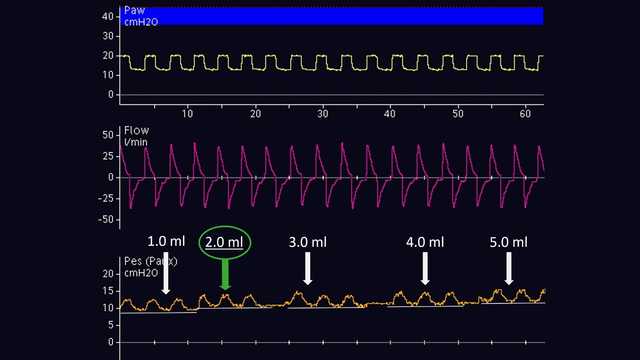
The volume of air for adequate inflation of the balloon should be individually titrated. This is only possible in passive patients. According to the method proposed by Mojoli et al (2016), the balloon is inflated from 0.5 to 3 ml in gradual steps of 0.5 ml for a Cooper Surgical catheter, and from 1 to 8 ml in steps of 1 ml for a Nutrivent catheter (see Figure 6). During progressive inflation of the balloon, the baseline of esophageal pressure increases and the magnitude of the esophageal pressure deflection changes. The adequate inflation volume is the one associated with the largest deflection of esophageal pressure. If two different inflation volumes show the same magnitude of esophageal pressure deflection, the lowest inflation volume is selected.
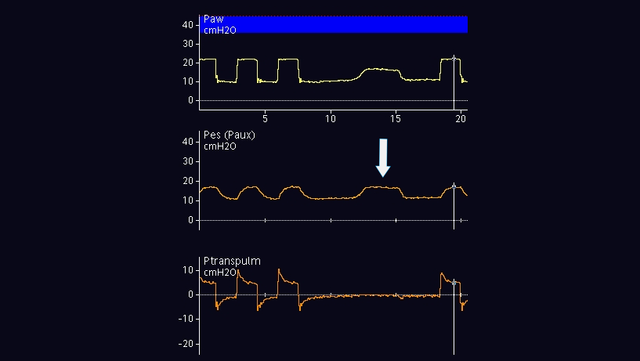
Once the balloon has been positioned correctly in the esophagus and inflated, verification is performed by means of an occlusion test. The principle is to close the airways at end expiration to change the airway pressure, and then to verify that esophageal pressure changes by the same amount.
In passive patients, you can perform an end-expiratory occlusion. When the expiratory valve is closed, apply an external manual compression of the rib cage on both sides of the chest in order to see a positive deflection of airway and esophageal pressures. The magnitude of the increase in airway and esophageal pressure should be the same. In other words, the transpulmonary pressure should not change (see Figure 7).
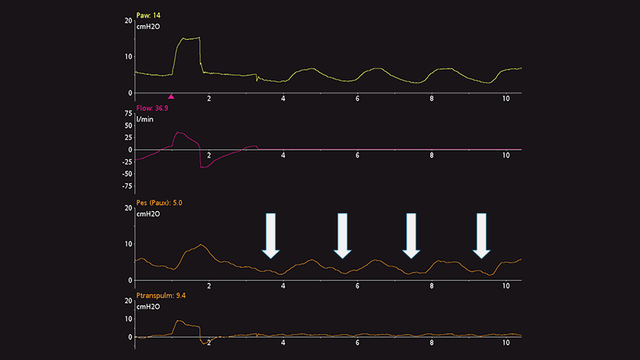
In active patients, the dynamic occlusion test also uses an end-expiratory occlusion. There is no need to press manually on the chest, as the patient will make a spontaneous inspiratory effort during the occlusion. The result is a negative deflection of airway and esophageal pressure. The magnitude of the decrease in airway and esophageal pressure should be the same, i.e., the transpulmonary pressure should not change (see Figure 8).
If you wish to monitor esophageal pressure continuously, it is important to reassess the correct position and inflation volume.
Full citations below: (

Clinically proven recommendations about what to do and what to avoid when using esophageal pressure in ARDS patients.